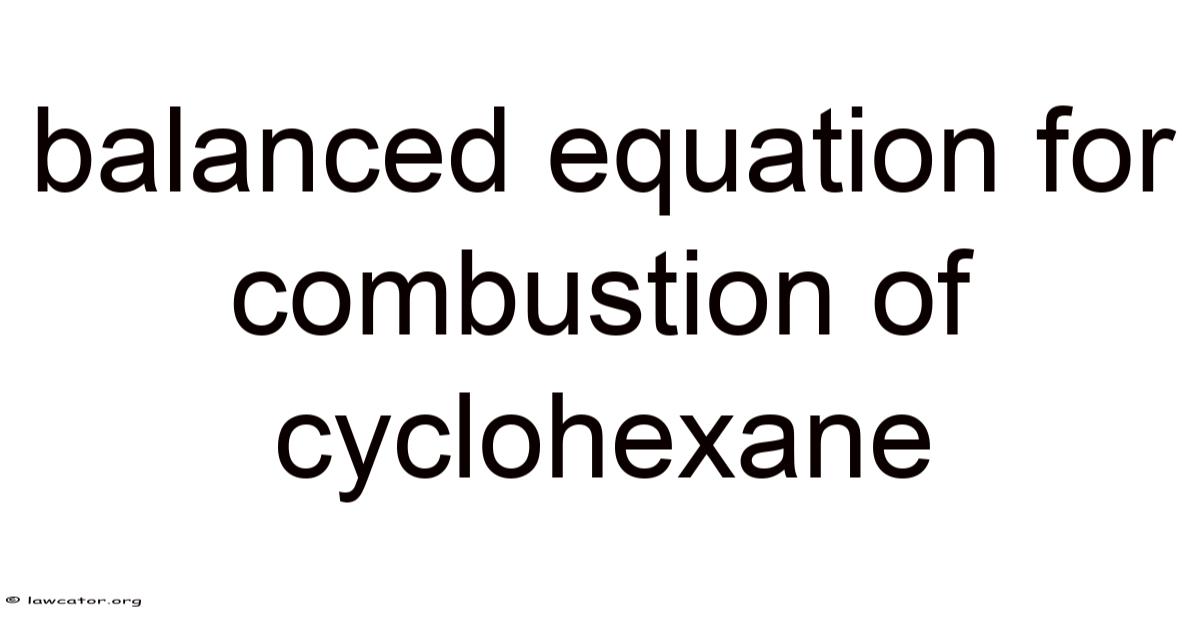Balanced Equation for Combustion of Cyclohexane: A Complete Guide
The combustion of cyclohexane is a fundamental chemical reaction that exemplifies the energy release from hydrocarbon fuels. Think about it: understanding its balanced equation is crucial for students of chemistry, environmental science, and engineering, as it provides insight into stoichiometry, energy production, and the environmental impact of burning fossil fuel derivatives. This reaction, at its core, is a high-temperature, exothermic oxidation process where cyclohexane reacts with oxygen to produce carbon dioxide, water, and heat Took long enough..
The Core Balanced Chemical Equation
The foundation of this topic is the balanced chemical equation for the complete combustion of liquid cyclohexane (C₆H₁₂). The unbalanced reaction is straightforward:
C₆H₁₂ (l) + O₂ (g) → CO₂ (g) + H₂O (g)
On the flip side, this equation violates the Law of Conservation of Mass, which states that matter cannot be created or destroyed in a chemical reaction. The number of atoms for each element must be equal on both sides of the equation. Balancing it requires adjusting the coefficients—the numbers placed in front of the chemical formulas Small thing, real impact..
The correct, balanced equation is:
2 C₆H₁₂ (l) + 19 O₂ (g) → 12 CO₂ (g) + 12 H₂O (g)
This equation tells us that two molecules of liquid cyclohexane require nineteen molecules of oxygen gas to undergo complete combustion, yielding twelve molecules of carbon dioxide and twelve molecules of water vapor. The heat released (ΔH) is substantial, characteristic of hydrocarbon combustion.
Step-by-Step Balancing Process
Balancing this equation is an excellent exercise in stoichiometry. Here is the systematic approach:
-
Balance Carbon (C) atoms first. There are 6 carbon atoms in one C₆H₁₂ molecule. To balance, place a coefficient of 12 in front of CO₂ Worth keeping that in mind..
- Unbalanced: C₆H₁₂ + O₂ → 12 CO₂ + H₂O
-
Balance Hydrogen (H) atoms next. There are 12 hydrogen atoms in one C₆H₁₂ molecule. Each H₂O molecule contains 2 hydrogens, so you need 6 H₂O molecules to balance the hydrogens from one cyclohexane No workaround needed..
- Unbalanced: C₆H₁₂ + O₂ → 12 CO₂ + 6 H₂O
-
Finally, balance Oxygen (O) atoms. This is often the trickiest step. On the product side, calculate total oxygen atoms:
- From 12 CO₂: 12 molecules * 2 O atoms = 24 O atoms
- From 6 H₂O: 6 molecules * 1 O atom = 6 O atoms
- Total O atoms on right = 30
- Each O₂ molecule provides 2 oxygen atoms. To get 30 O atoms, you need 15 O₂ molecules (15 * 2 = 30).
- Unbalanced: C₆H₁₂ + 15 O₂ → 12 CO₂ + 6 H₂O
-
Adjust for Whole Number Coefficients. The equation is now balanced for atoms, but we typically want the smallest set of whole-number coefficients. The coefficients (1, 15, 12, 6) have a greatest common divisor of 1, so they are already the simplest form. Even so, to avoid fractions in more complex balancing, we often start with a fractional coefficient. Let's verify: If we used 1 C₆H₁₂, we'd need 9.5 O₂, which is not ideal. Multiplying the entire equation by 2 to clear the fraction (from the H balance) gives us the final, standard form:
- 2 C₆H₁₂ + 19 O₂ → 12 CO₂ + 12 H₂O
The Science Behind the Flames: Complete vs. Incomplete Combustion
The balanced equation above represents complete combustion, which occurs when ample oxygen is present. On the flip side, the products are always carbon dioxide (CO₂) and water (H₂O). This reaction is highly exothermic, releasing approximately -3920 kJ/mol of energy, making cyclohexane a potent fuel.
In real-world scenarios, oxygen is often limited. This leads to incomplete combustion, a dangerous process that produces carbon monoxide (CO), a colorless, odorless, and highly toxic gas, along with carbon (soot) and water. The equations for incomplete combustion of cyclohexane can vary:
- For Carbon Monoxide and Water:
- 2 C₆H₁₂ + 17 O₂ → 12 CO + 12 H₂O
- For Carbon (Soot) and Water:
- 2 C₆H₁₂ + 11 O₂ → 12 C + 12 H₂O
These variations highlight why ensuring proper ventilation for any combustion process—from car engines to home heating systems—is a critical safety issue But it adds up..
Practical Applications and Importance
Understanding this balanced equation extends far beyond the classroom:
- Energy Calculations: The stoichiometry allows for precise calculation of the amount of energy released from a given mass of cyclohexane, vital for fuel efficiency analysis.
- Environmental Impact: By knowing that 2 moles of cyclohexane produce 12 moles of CO₂, we can calculate the exact carbon footprint of burning a specific quantity. This is fundamental to carbon emissions accounting.
- Stoichiometric Air-Fuel Ratios: In engineering, the equation is used to determine the theoretical air required for complete combustion. The molar ratio of O₂ to C₆H₁₂ is 19:2, or 9.5:1. This is scaled up using air's composition (~21% O₂) to find the ideal air-to-fuel mass ratio for combustion engines.
- Safety Protocols: Recognizing the products of incomplete combustion underscores the necessity of carbon monoxide detectors and proper appliance maintenance.
Frequently Asked Questions (FAQ)
Q: Why is it important to balance the chemical equation? A: Balancing ensures the equation obeys the Law of Conservation of Mass. It provides the correct mole ratios, which are essential for all quantitative predictions in chemistry, such as calculating reactant needs, product yields, and energy changes No workaround needed..
Q: Is cyclohexane combustion the same as for other hydrocarbons like hexane or benzene? A: The general pattern is the same: hydrocarbon + oxygen → carbon dioxide + water. Even so, the coefficients differ based on the hydrocarbon's specific C:H ratio. Here's one way to look at it: the balanced equation for complete combustion of n-hexane (C₆H₁₄) is: 2 C₆H₁₄ + 19 O₂ → 12 CO₂ + 14 H₂O. Benzene (C₆H₆) is: 2 C₆H₆ + 15 O₂ → 12 CO₂ + 6 H₂O.
Q: What does the state symbol (l) or (g) mean in the equation? A: The (l) denotes the physical state of cyclohexane under standard conditions (25°C, 1 atm) as a liquid. (g) indicates gases (oxygen, carbon dioxide, water vapor). In an actual flame, water is initially produced as hot vapor (steam) Simple, but easy to overlook..
Q: How does this equation relate to climate change? A: The 12 moles of CO₂ produced from
from 2 moles of cyclohexane directly contribute to the greenhouse effect, trapping heat in the atmosphere and driving global warming. This makes cyclohexane, like all fossil fuels, a significant contributor to climate change when combusted. The sheer volume of hydrocarbons burned globally for energy and industrial processes means even small imbalances in combustion efficiency have large-scale environmental consequences Still holds up..
Beyond that, the potential for incomplete combustion underscores the broader environmental impact. Soot (particulate carbon) contributes to air pollution, respiratory problems, and climate change by absorbing sunlight and darkening surfaces (reducing albedo). Carbon monoxide (CO) is a toxic gas harmful to human health and ecosystems. These secondary pollutants highlight the critical importance of optimizing combustion conditions not just for efficiency, but for minimizing harmful outputs.
Conclusion
The balanced chemical equation for the combustion of cyclohexane, C₆H₁₂(l) + 9 O₂(g) → 6 CO₂(g) + 6 H₂O(g), is far more than a simple representation of a chemical reaction. That said, it is a fundamental tool that bridges theoretical chemistry with real-world applications. On the flip side, understanding its stoichiometry allows for precise energy calculations, accurate environmental impact assessments, and the design of efficient combustion systems. But crucially, it vividly illustrates the delicate balance required for complete combustion and the serious consequences of imbalance, including the production of life-threatening carbon monoxide and environmentally damaging soot. This knowledge underscores the critical importance of engineering solutions, safety protocols, and environmental stewardship in managing the combustion of hydrocarbons, ensuring both operational efficiency and the protection of human health and the planet for future generations Small thing, real impact..