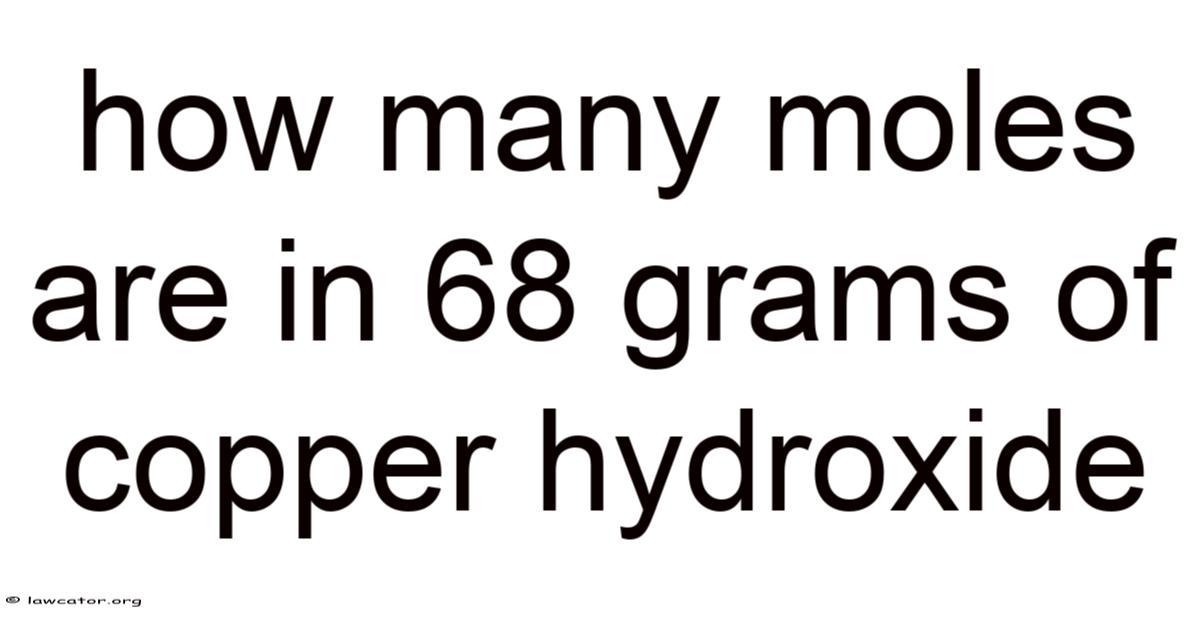The precise calculation of moles serves as a foundational pillar in understanding chemical compositions, particularly when dealing with substances like copper hydroxide whose properties underpin countless applications across scientific disciplines. Even so, whether one is conducting a lab experiment, analyzing environmental samples, or designing industrial processes, grasping the relationship between mass, molar mass, and molecular quantity is essential. Copper hydroxide, commonly denoted as Cu(OH)₂, presents both theoretical and practical challenges that demand meticulous attention to detail. Which means its unique chemical structure, composed of copper ions bound to hydroxide ions, introduces complexities that necessitate careful consideration of atomic weights, stoichiometric ratios, and precise measurements. In this context, the task of determining how many moles exist within a given mass of copper hydroxide becomes not merely a numerical exercise but a critical step toward achieving desired outcomes in various scientific endeavors. Also, such precision underscores the importance of foundational knowledge in chemistry, where even minor inaccuracies can cascade into significant consequences, emphasizing the necessity of thorough preparation and careful execution. In practice, the process itself offers opportunities to explore the interplay between macroscopic measurements and microscopic principles, revealing how fundamental concepts like molar mass translate abstract quantities into actionable data. Beyond its utility in calculations, this task also serves as a gateway to deeper engagement with the subject matter, inviting learners to ponder broader implications of their work. It bridges the gap between theoretical understanding and practical application, ensuring that the abstract principles remain anchored in tangible reality. But this foundational task, while seemingly straightforward on the surface, demands a meticulous approach that reflects the discipline’s inherent rigor, reinforcing the value of precision in scientific inquiry. The act of converting grams into moles, for instance, requires not only mathematical accuracy but also an awareness of unit consistency and the underlying assumptions that govern the process. Such attention ensures that subsequent steps—whether determining concentration, calculating reactions, or interpreting results—are grounded in reliable data. In this light, the calculation of moles becomes a microcosm of scientific methodology itself, illustrating how foundational steps permeate the entire field of chemistry, influencing everything from lab protocols to theoretical models. Day to day, the process invites reflection on the iterative nature of scientific work, where repeated verification and adjustment are often necessary to attain consensus or achieve desired outcomes. To build on this, the significance of this calculation extends beyond immediate calculations; it underpins more complex analyses that rely on accurate baseline data. Whether assessing solubility, tracking chemical equilibria, or evaluating material properties, the number of moles acts as a universal bridge connecting disparate aspects of a system. In real terms, understanding its role necessitates not only computational proficiency but also a nuanced grasp of the subject’s broader context, ensuring that each step is purposeful and purposeful. Because of that, this interconnection between individual calculations and larger-scale applications highlights the multifaceted nature of scientific work, where precision at the micro level supports precision at the macro level. The importance of this task becomes evident when considering real-world scenarios where even small errors can lead to substantial discrepancies, necessitating rigorous adherence to standards That alone is useful..
In such contexts, the meticulous application of fundamental principles becomes not merely a procedural formality but a cornerstone of credible scientific practice. Researchers and students alike must recognize that each calculation, no matter how elementary it may appear, contributes to a larger framework of knowledge that informs everything from pharmaceutical development to environmental monitoring.
Most guides skip this. Don't.
The journey from understanding mole concepts to executing precise conversions embodies the essence of scientific literacy. Even so, it cultivates habits of mind—attention to detail, skepticism toward unverified assumptions, and commitment to reproducibility—that transcend the specific subject matter and become transferable skills applicable across disciplines. When one masters the art of translating between grams and moles, they are simultaneously developing a deeper appreciation for the quantitative nature of the physical world Took long enough..
As we reflect on the broader educational implications, it becomes clear that such foundational exercises serve dual purposes: they equip learners with practical computational tools while simultaneously instilling an appreciation for the elegance underlying chemical principles. The mole, in many ways, represents a triumph of scientific abstraction—a means of making the invisible measurable and the infinitesimal comprehensible.
Not obvious, but once you see it — you'll see it everywhere.
Pulling it all together, the calculation of moles from a given mass is far more than a routine exercise in stoichiometry; it is a gateway to understanding the language of chemistry itself. By approaching such tasks with the seriousness they deserve, we not only ensure accuracy in our immediate work but also contribute to a culture of scientific rigor that benefits the entire enterprise of discovery. The small steps we take in mastering these fundamentals ultimately pave the way for the great strides that define scientific progress Easy to understand, harder to ignore..
This changes depending on context. Keep that in mind Most people skip this — try not to..
Continuing the Article on Mole Calculations and Scientific Practice
The journey from understanding mole concepts to executing precise conversions embodies the essence of scientific literacy. Which means it cultivates habits of mind—attention to detail, skepticism toward unverified assumptions, and commitment to reproducibility—that transcend the specific subject matter and become transferable skills applicable across disciplines. When one masters the art of translating between grams and moles, they are simultaneously developing a deeper appreciation for the quantitative nature of the physical world.
It sounds simple, but the gap is usually here.
As we reflect on the broader educational implications, it becomes clear that such foundational exercises serve dual purposes: they equip learners with practical computational tools while simultaneously instilling an appreciation for the elegance underlying chemical principles. The mole, in many ways, represents a triumph of scientific abstraction—a means of making the invisible measurable and the infinitesimal comprehensible No workaround needed..
In practice, these skills manifest in numerous professional contexts. Pharmaceutical chemists rely on precise molar calculations to determine correct dosages and drug formulations, ensuring both efficacy and patient safety. Because of that, environmental scientists use these same principles to assess pollutant concentrations in air and water, informing policy decisions that protect public health. Materials engineers apply stoichiometric reasoning to synthesize new compounds with tailored properties, driving innovation across industries from electronics to renewable energy Still holds up..
The pedagogical value of mastering such calculations extends beyond the chemistry laboratory. Students who develop fluency in dimensional analysis and unit conversion carry these competencies into physics, biology, economics, and engineering. The analytical frameworks developed through chemical calculations—breaking complex problems into manageable steps, verifying results through multiple methods, and understanding the significance of significant figures—prepare learners for the quantitative reasoning demanded in virtually every STEM field It's one of those things that adds up. Nothing fancy..
Also worth noting, the process of working through mole calculations reinforces a fundamental truth about scientific inquiry: knowledge advances through incremental accumulation. And each correctly solved problem adds to a foundation upon which more complex understanding can be built. Just as a architect cannot construct a skyscarter without first mastering the principles of load-bearing, scientists cannot tackle advanced problems without fluency in foundational calculations.
The interconnected nature of scientific knowledge becomes particularly apparent when considering how mole calculations connect to broader theoretical frameworks. Understanding why one mole of any substance contains exactly 6.Avogadro's number, far from being an arbitrary constant, represents a profound insight into the relationship between the macroscopic and microscopic worlds. 022 × 10²³ entities illuminates the deep symmetries underlying chemical behavior and prepares students to appreciate more sophisticated concepts like molecular dynamics, thermodynamic relationships, and quantum mechanical models.
Counterintuitive, but true.
In educational settings, instructors bear the responsibility of presenting these concepts not as isolated procedures but as part of a coherent intellectual tradition. When students understand why formulas work—not merely how to apply them—they become better equipped to handle novel situations where standard approaches may not suffice. This conceptual depth transforms rote memorization into genuine comprehension, fostering the kind of flexible thinking that characterizes expert problem-solving.
The assessment of computational skills also warrants careful consideration. Effective evaluation goes beyond testing whether students can produce correct numerical answers; it should also examine whether learners understand the reasoning behind their calculations, can identify potential sources of error, and know how to verify their results through independent methods. Such comprehensive assessment encourages deeper learning and provides more meaningful feedback about student understanding Simple as that..
Worth pausing on this one Easy to understand, harder to ignore..
Looking toward the future, advances in computational chemistry and artificial intelligence will undoubtedly transform how chemical calculations are performed in research settings. On the flip side, the fundamental principles underlying these calculations will remain essential. So scientists must understand the basis of computational tools to use them appropriately, recognize when results are physically reasonable, and effectively interpret the outputs of sophisticated modeling software. Thus, foundational skills in mole calculations and stoichiometry will remain relevant even as specific computational methods evolve It's one of those things that adds up..
To build on this, the democratization of scientific knowledge through open-access publications, educational resources, and citizen science initiatives places increased emphasis on quantitative literacy among non-specialists. Also, members of the public who wish to evaluate scientific claims, participate in policy discussions, or contribute to research projects will benefit from understanding fundamental concepts like molar relationships. This broader social dimension underscores the importance of effective science education that goes beyond training specialists to cultivating an informed citizenry.
Not obvious, but once you see it — you'll see it everywhere Not complicated — just consistent..
At the end of the day, the calculation of moles from a given mass is far more than a routine exercise in stoichiometry; it is a gateway to understanding the language of chemistry itself. Day to day, by approaching such tasks with the seriousness they deserve, we not only ensure accuracy in our immediate work but also contribute to a culture of scientific rigor that benefits the entire enterprise of discovery. The small steps we take in mastering these fundamentals ultimately pave the way for the great strides that define scientific progress.
Real talk — this step gets skipped all the time Worth keeping that in mind..