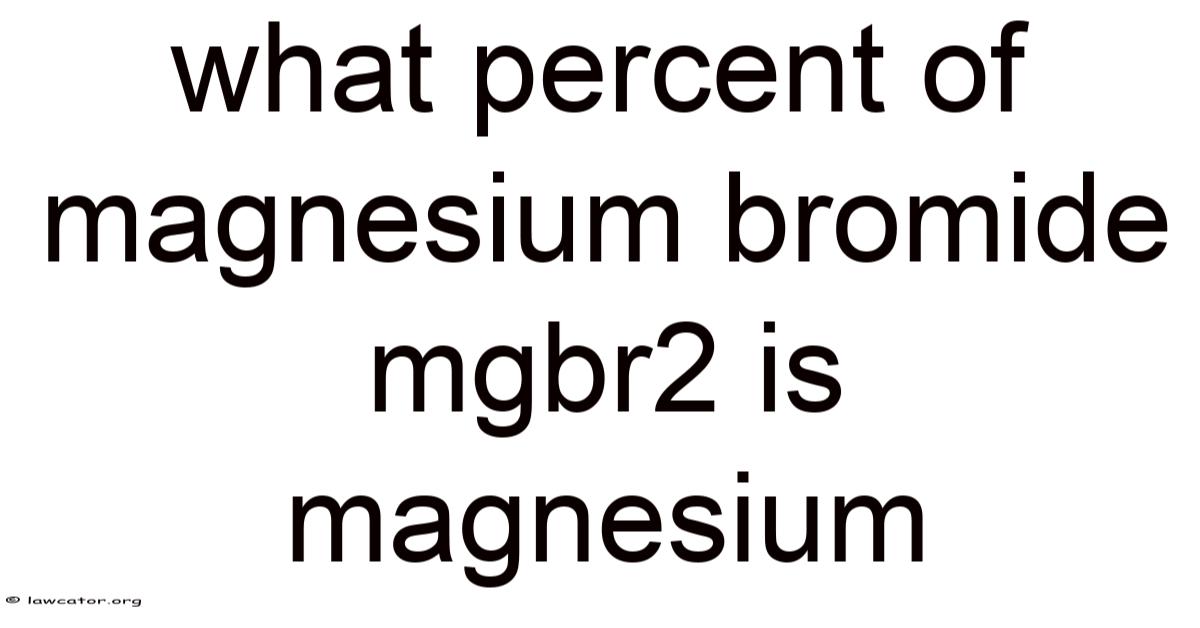What Percent of Magnesium Bromide (MgBr₂) Is Magnesium?
Magnesium bromide (MgBr₂) is a chemical compound composed of magnesium (Mg) and bromine (Br) atoms. * To answer this, we need to calculate the mass contribution of magnesium relative to the total molar mass of the compound. Understanding its composition is essential in chemistry, as it helps determine the proportions of elements within the compound. One common question in this context is: *What percentage of magnesium bromide is magnesium?This process involves basic stoichiometric principles and is a foundational skill in chemistry It's one of those things that adds up..
Understanding Percent Composition
Percent composition refers to the percentage by mass of each element in a chemical compound. It is calculated using the formula:
$
\text{Percent Composition} = \left( \frac{\text{Mass of Element}}{\text{Molar Mass of Compound}} \right) \times 100
$
For magnesium bromide (MgBr₂), we need to determine the molar mass of the compound and the mass of magnesium within it. This allows us to quantify how much of the compound’s total mass is attributable to magnesium Most people skip this — try not to..
Step 1: Determine the Molar Mass of MgBr₂
The molar mass of a compound is the sum of the atomic masses of all atoms in its formula. Using the periodic table:
- Magnesium (Mg): 24.305 g/mol
- Bromine (Br): 79.904 g/mol
Since there are two bromine atoms in MgBr₂, their combined mass is:
$
2 \times 79.904 = 159.808 , \text{g/mol}
$
Adding magnesium’s mass gives the total molar mass of MgBr₂:
$
24.305 + 159.808 = 184 That's the part that actually makes a difference..
This changes depending on context. Keep that in mind.
Step 2: Calculate the Mass Contribution of Magnesium
Magnesium contributes 24.305 g/mol to the total molar mass of MgBr₂. To find its percentage:
$
\text{Percent Magnesium} = \left( \frac{24.305}{184.113} \right) \times 100
$
Performing the division:
$
\frac{24.305}{184.113} \approx 0.132
$
Multiplying by 100 converts this to a percentage:
$
0.132 \times 100 = 13.2%
$
Step 3: Interpret the Result
The calculation shows that 13.2% of magnesium bromide’s mass comes from magnesium atoms. The remaining 86.8% is due to bromine. Basically, in every 100 grams of MgBr₂, 13.2 grams are magnesium, and 86.8 grams are bromine And that's really what it comes down to. That's the whole idea..
This result is critical for applications such as:
- Chemical synthesis: Ensuring precise ratios of elements in reactions.
In practice, - Analytical chemistry: Identifying unknown compounds by their elemental composition. - Industrial processes: Optimizing the use of raw materials in manufacturing.
Why Does This Matter?
Knowing the percent composition of compounds like MgBr₂ is vital in both academic and industrial settings. Take this: in pharmaceuticals, accurate elemental proportions ensure drug efficacy and safety. In environmental science, understanding compound composition helps assess the impact of chemicals on ecosystems.
Additionally, this calculation reinforces the concept of stoichiometry, which governs the quantitative relationships in chemical reactions. It also highlights the importance of atomic masses in determining molecular structure and behavior.
Common Mistakes to Avoid
When calculating percent composition, students often make errors such as:
- Using incorrect atomic masses: Always verify values from a reliable periodic table.
- Miscounting atoms: Double-check the subscript in the chemical formula (e.g., MgBr
Understanding the mass distribution within magnesium bromide not only reinforces fundamental chemistry principles but also equips scientists and engineers with precise data for practical applications. Because of that, by systematically analyzing the molar masses and percentage contributions, we gain deeper insights into how elements interact in compounds. This exercise underscores the significance of accuracy in scientific work, whether in lab experiments or industrial formulations.
To wrap this up, the analysis of magnesium’s role in MgBr₂ illustrates the interplay between atomic weights and mass percentages, offering a clear pathway to master quantitative chemistry. Such calculations are indispensable tools for anyone aiming to excel in the field Nothing fancy..
Conclusion: Mastering these concepts enhances analytical skills and ensures reliability in chemical research and real-world problem-solving.
- Neglecting to multiply by 100: After dividing the element’s mass by the molar mass of the compound, remember to convert the decimal to a percent.
- Ignoring significant figures: Report the final percentage with the appropriate number of significant digits based on the precision of the atomic masses used.
- Forgetting to account for all atoms in the formula: In MgBr₂ there are two bromine atoms; omitting one leads to an incorrect total molar mass and a skewed percent composition.
By being mindful of these pitfalls, you’ll obtain accurate and reproducible results every time.
Applying Percent Composition in Real‑World Scenarios
- Pharmaceutical formulation: When preparing a magnesium‑based antacid, knowing that only ~13 % of MgBr₂ is magnesium helps pharmacists calculate the exact amount of active ingredient needed per tablet.
- Materials science: In the production of flame‑retardant polymers, the bromine content (≈ 87 %) determines the compound’s effectiveness and influences the required loading levels.
- Environmental monitoring: Detecting trace amounts of magnesium bromide in water samples relies on precise percent‑composition data to quantify pollutant concentrations.
These examples illustrate how a simple percent‑composition calculation translates into actionable information across diverse fields.
Conclusion
Determining that magnesium constitutes about 13 % of magnesium bromide’s mass exemplifies the power of stoichiometric analysis. Accurate percent composition not only ensures the reliability of laboratory results but also informs decision‑making in industrial processes and environmental assessments. In real terms, by mastering this calculation, you gain a foundational skill that underpins more complex quantitative work in chemistry, engineering, and applied sciences. Embrace these calculations as essential tools in your scientific toolkit, and you’ll be well‑equipped to tackle a wide range of chemical challenges with confidence.
Extending Percent Composition Skills to Other Compounds
The methodology demonstrated with MgBr₂ applies universally to any chemical compound. Here's one way to look at it: calculating the percent composition of calcium carbonate (CaCO₃) follows the same steps: determine the molar mass of the compound, identify the mass contribution of each element, and express these as percentages. This approach is equally valuable for complex molecules like glucose (C₆H₁₂O₆) or hydrated salts such as copper(II) sulfate pentahydrate (CuSO₄·5H₂O) Turns out it matters..
Practice Problem: Try calculating the percent composition of aluminum nitrate, Al(NO₃)₃. You'll find that nitrogen comprises approximately 26 % of the compound's mass, while oxygen accounts for roughly 53 %. These exercises reinforce the pattern recognition essential for rapid, accurate calculations in both academic and professional settings.
Digital Tools and Computational Aids
Modern chemistry education increasingly incorporates computational tools to verify manual calculations. Always perform the calculation by hand first, then use technology to confirm your results. And spreadsheet software, molecular modeling apps, and online calculators can cross-check your work, but understanding the underlying mathematics remains crucial. This dual approach builds intuition while ensuring accuracy Not complicated — just consistent. That's the whole idea..
Final Thoughts
Percent composition calculations serve as a gateway to deeper chemical understanding. They connect abstract atomic theory to tangible, measurable quantities, bridging the gap between molecular structure and macroscopic properties. Whether you're analyzing the magnesium content in a dietary supplement or determining the bromine levels in industrial waste, these skills prove indispensable.
Mastering percent composition isn't just about memorizing formulas—it's about developing a chemist's mindset that sees beyond the numbers to the molecular reality they represent. With practice and attention to detail, anyone can become proficient in this fundamental aspect of chemical analysis.
Final Conclusion
The journey from understanding atomic masses to calculating percent composition in compounds like MgBr₂ represents more than a mathematical exercise—it embodies the essence of chemical thinking. Think about it: by internalizing these concepts and avoiding common calculation pitfalls, you equip yourself with tools that extend far beyond the classroom. Whether in pharmaceutical development, materials engineering, or environmental science, the ability to quantify elemental composition remains a cornerstone of scientific literacy. Here's the thing — as you continue your studies or professional work, remember that every complex problem begins with these fundamental building blocks. Embrace them, practice them, and watch how they illuminate the invisible world of atoms and molecules that surrounds us.